 
It can sometimes lose two electrons to form the Fe 2 + ion, while at other times it loses three electrons to. This pattern continues, meaning that elements in group 7 have ions. This is because the elements in group 1 have one valence electron and those in group 2 have two valence electrons. An ion ( / a.n, - n /) 1 is an atom or molecule with a net electrical charge. Forming an ionic bond, Li and F become Li + and F ions. Elements in group 2 will have a charge of +2. Ion Electron transfer between lithium (Li) and fluorine (F). As an example, iron commonly forms two different ions. Elements in group 1 (that is the first column of the periodic table) will usually have ions which have a +1 charge. An iron ion with a charge of +8 is not very likely, therefore, the octet rule is not applicable to transition elements. Most transition metals differ from the metals of Groups 1, 2, and 13 in that they are capable of forming more than one cation with different ionic charges. In order for an element such as iron (Fe) to achieve the same noble gas configuration of argon (Ar), it would need to lose 6 electrons in the 3 d subshell and 2 electrons in the 4s subshell. Each element square contains all 118 of elements with the element number, symbol, name, atomic mass, and most common oxidation number. Reviewing the way of finding the charge on an atom may help, too. 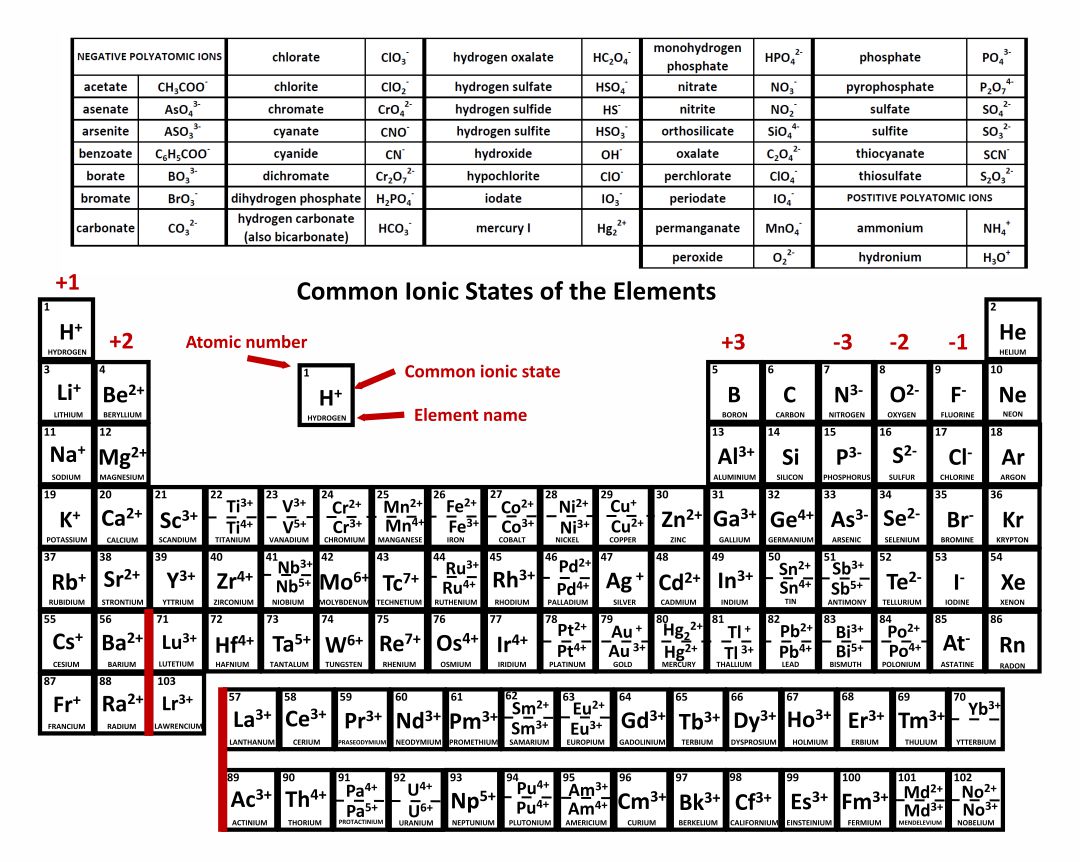
This is because the transition metals have electrons in d subshell and do not follow the octet rule. The periodic table ionic charge can be broken down by metals that are positive and on the left of the table and nonmetals which are negative and found on. This periodic table with charges is a useful way to keep track of the most common oxidation numbers for each element. Notice that there is no simple pattern for transition metal ions (or for the larger main group elements) as there is with the main group ions. \) shows the characteristic charges for some of these ions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
